The excellent characteristics of lithium-ion batteries
Lithium-ion batteries serve as the power source for devices like smartphones and laptops. With advancements in technology, these batteries have become smaller in size and larger in capacity, and are now also used in drones and electric bicycles. Lithium-ion batteries can be considered indispensable in our daily lives. They are widely utilized across various fields due to the following three main advantages:
- High voltage and high energy density
The voltage of lithium-ion batteries (nominal voltage of 3.7 V) is approximately three times that of nickel-cadmium (NiCd) or nickel-metal hydride (NiMH) batteries of the same size (both with a nominal voltage of 1.2 V). This means that compared to traditional batteries, they can produce the same voltage with fewer battery cells. Additionally, the energy density of lithium-ion batteries is higher than that of other batteries, allowing products to be smaller and lighter. - Self-discharge characteristics
The self-discharge rate of lithium-ion batteries is approximately 5% per month, which is less than one-fifth of the self-discharge rate of nickel-cadmium or nickel-metal hydride batteries. This means that even if the battery is left idle for several months, it can still retain energy comparable to what traditional batteries would have after just one month of idle time. - No memory effect
In traditional nickel-cadmium and nickel-metal hydride batteries, repeatedly performing partial charging can reduce the overall battery lifespan, a phenomenon known as the 'memory effect.' Lithium-ion batteries, however, do not have this memory effect, allowing them to be recharged repeatedly.

The safety of lithium-ion batteries
The diagram below illustrates the classification of batteries. This article focuses on rechargeable batteries (batteries that can be recharged and reused), which fall under the category of chemical batteries. Lithium-ion batteries are a type of high-performance battery that can be miniaturized and made lightweight, with performance that far surpasses traditional batteries used in electronic devices. Despite the many advantages of lithium-ion batteries, there are still some challenges, such as the risk of overcharging.

For example, the following are the active materials for the positive and negative electrodes, as well as the electrolytes, that make up rechargeable batteries (lead-acid batteries, nickel-cadmium batteries, nickel-metal hydride batteries, and lithium-ion batteries). Among the electrolytes, all batteries use water as the solvent except for lithium-ion batteries, which use organic solvents. This is intended to achieve a nominal voltage exceeding the voltage of water electrolysis (1.23 V).

When nickel-metal hydride batteries or nickel-cadmium batteries are being charged, the battery voltage gradually increases, reaching a peak as it approaches full charge, and then stabilizes. At the same time, the battery temperature also continues to rise, but the increase is not significant. Although the battery capacity is relatively small compared to its size, these types of batteries are relatively safe because the electrolyte is an aqueous solution (non-flammable).
In comparison, if a lithium-ion battery continues to charge without any protective measures, even after the battery is fully charged, the battery voltage and temperature will continue to rise (see the diagram below).
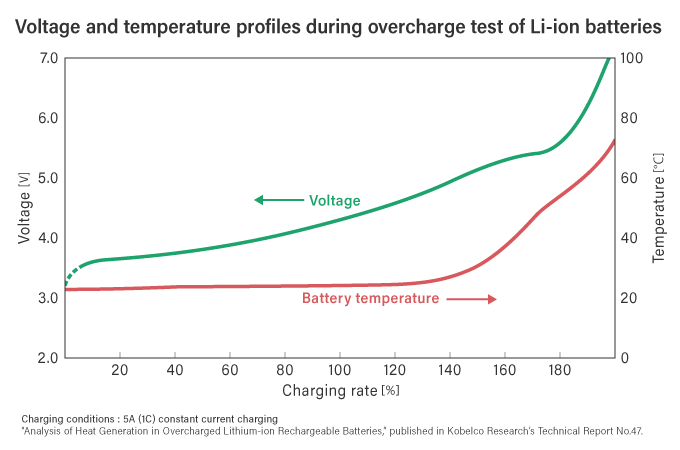
In a confined space storing a large amount of energy under high-temperature conditions, combined with the presence of flammable organic solvents (flammable liquids), these factors indeed make it a type of battery where 'overcharging can be dangerous.' Nevertheless, the undeniable advantages of such batteries, including their high energy density and other excellent characteristics, cannot be overlooked. Years of technological improvements and enhanced safety measures have contributed to their widespread adoption in the market.
Secondary protection of lithium-ion batteries
Rechargeable batteries are all equipped with a Battery Management System (BMS), which is located between the battery and the device (or charger) and is responsible for managing the charging and discharging processes. Primary protection is typically achieved through semiconductors that continuously monitor current, voltage, and temperature, and protection control is carried out via electronic circuits. Most abnormal conditions can be prevented through primary protection.
However, in very rare cases, semiconductors may fail to operate normally due to abnormalities or malfunctions, resulting in the failure of protective functions. Abnormalities in lithium-ion batteries can lead to severe consequences such as rupture or fire. Therefore, it is essential to preemptively consider protective measures for "in case of failure." As a result, a secondary protection mechanism has been implemented, with higher reliability requirements to ensure that it can perform its protective function under any circumstances.
Self-Control Protector (SCP)
The 'Self-Control Protector' (SCP) developed by Dexerials is a fuse component that provides secondary protection for lithium-ion batteries by irreversibly and physically disconnecting the charge and discharge circuit.
When the SCP is activated, it usually indicates that the primary protection (BMS) has failed and the overall condition of the lithium-ion battery has become abnormal. Therefore, the function of the SCP is to actively disconnect the circuit to prevent the unstable lithium-ion battery from continuing to operate, ensuring safety.
Generally speaking, traditional fuses physically disconnect the circuit by melting the fuse element through Joule heating, preventing equipment damage caused by overcurrent. However, in addition to overcurrent protection, lithium-ion batteries also require consideration of overcharging scenarios. SCP was developed specifically to address this need. Since its launch in 1994, SCP has been recognized as the standard component for secondary protection in lithium-ion batteries. As of March 2020, its shipment volume has exceeded 2 billion units.
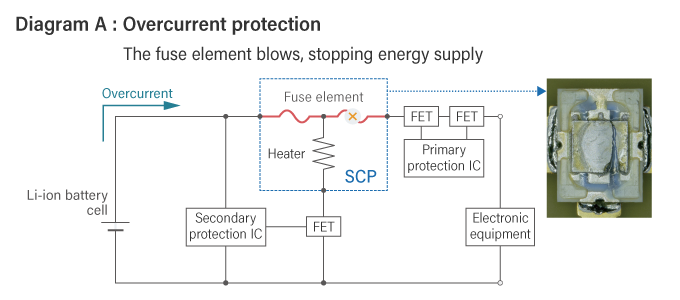
The structure and circuit diagram of the SCP are shown below.

The operation of SCP during overcurrent is similar to that of a traditional fuse, where the fuse element melts and breaks the circuit. However, SCP has a unique activation method during overcharging. When overcharging is detected, the heater located beneath the fuse element in the SCP activates and melts the fuse element, thereby cutting off the current.
The diagram A below shows the operation of the SCP during overcurrent, while diagram B shows the operation of the SCP during overcharging.

The Potential and Future of SCP
For approximately 30 years, Dexerials has been dedicated to the research and development of lithium-ion battery protection. Its proprietary SCP technology has been widely used as a secondary protection device for lithium-ion batteries, achieving a 60-70% market share in the laptop market and becoming an important industry benchmark.
- Devices such as smartphones or tablets:
SCP plays a critical role in these devices by preventing battery overcharging or over-discharging, extending battery life, and enhancing safety, thereby significantly improving user experience. - Power tools such as electric tools:
In the field of power tools, where durability and safety are of utmost importance, SCP serves as a critical safety component, ensuring that batteries operate stably even in harsh environments while maintaining high performance. - Drone:
As the market expands and technology innovates, the demand for lightweight and highly efficient battery management in drones is increasing. SCP not only ensures flight safety but also helps maximize flight time. - Electric cars and electric motorcycles:
In the field of electric vehicles, SCP enhances battery safety and enables longer driving ranges. It also has the capability to handle high currents, promoting the adoption of fast-charging technology.
SCP technology continues to evolve, and in the future, it will further develop towards 'miniaturization' and 'high current capacity' to create safer and more efficient lithium-ion batteries.

